BridgeBio Pharma Reports Fourth Quarter and Full Year 2019 Financial Results and Highlights Portfolio Progress
-Achieved multiple clinical and pre-clinical milestones across BridgeBio’s pipeline
-Delivered pipeline expansion with addition of five new drug development and discovery programs; BridgeBio’s portfolio now includes more than 20 potential medicines
-Ended quarter with $577.1 million in cash, cash equivalents and marketable securities
SAN FRANCISCO, March 02, 2020 (GLOBE NEWSWIRE) — BridgeBio Pharma, Inc. (NASDAQ: BBIO), a clinical-stage biopharmaceutical company focused on genetic diseases, today is reporting its fourth quarter and full year 2019 financial results and recent progress across its portfolio, which now includes more than 20 drug development and discovery programs.
2019 was a year of significant growth and milestone achievements for BridgeBio. The company initiated its first New Drug Application (NDA) with the United States Food and Drug Administration (FDA) and added five new drug development and discovery programs to its pipeline, which target diseases in endocrinology, ophthalmology, otology, neurology and musculoskeletal therapeutic areas. Veteran biotech and pharmaceutical industry leaders joined BridgeBio’s board of directors and the company as senior leaders, including the recent board appointment of Ronald J. Daniels, president of Johns Hopkins University. The company went public on June 27, 2019 and raised approximately $401 million in gross proceeds in its initial public offering.
“BridgeBio was founded nearly five years ago on the back of two big ideas: that there had to be a better way to finance critical biomedical research and that there was a tremendous amount of medical innovation that was trapped, without a way to move into the clinic where it could help patients,” said BridgeBio founder and CEO Neil Kumar, Ph.D. “Today BridgeBio has more than 20 drug development and discovery programs in our pipeline. We initiated our first new drug application with the FDA and we are on track to file our second later this year, along with the anticipated filing of multiple investigational new drug applications. We are demonstrating that our model is working and has the potential to produce an array of life-changing therapies for people in need.”
Pipeline growth:
We recently disclosed five new genetic medicine product candidates in the pipeline:
- Encaleret – Calcium sensing receptor antagonist for autosomal dominant hypocalcemia type 1 (ADH1): IND application for the treatment of ADH1 became effective in late 2019; Phase 2 ready.
- Zuretinol –Synthetic retinoid for inherited retinal disease due to RPE65 or LRAT gene mutations: Phase 2/3 study initiating in 2020.
- BBP-418 – Substrate supplementation therapy for limb-girdle muscular dystrophy type 2i (LGMD2i).
- BBP-472 – Brain-permeable inhibitor of PI3KBfor children with autism-spectrum disorders (ASD) characterized by loss of the PTEN protein. This program is in preclinical development.
- BBP-815 –AAV gene therapy for nonsyndromic hearing loss caused by recessive mutations in the TMC1 gene. This program is undergoing early proof of concept animal studies.
Fourth quarter 2019 and recent pipeline progress:
Mendelian
- Low-dose infigratinib – FGFR1-3 inhibitor for achondroplasia: Initiated clinical program consisting of an observational study (PROPEL, NCT04035811) and Phase 2 dose-ranging study (PROPEL2, NCT04265651) in children with achondroplasia, the most common form of genetic short stature.
- Fosdenopterin – cPMP replacement therapy for MoCD type A: Initiated a rolling submission of an NDA with the United States FDA for the treatment of patients with molybdenum cofactor deficiency (MoCD) type A.
- Topical patidegib gel for Gorlin syndrome and high-frequency basal cell carcinoma1: Completed enrollment of pivotal Phase 3 clinical trial in patients with Gorlin Syndrome (NCT03703310) and initiated Phase 2 study in high-frequency basal cell carcinoma (NCT04155190).
- BBP-265 (AG10) – TTR stabilizer for ATTR: Presented positive data from Phase 2 Open Label Extension (NCT03536767) in transthyretin amyloid cardiomyopathy at the American Heart Association’s 2019 Scientific Sessions.
___________
1 PellePharm, which is focused on developing patidegib topical gel, 2% entered into a strategic collaboration with LEO Pharma in November 2018, which includes an option for LEO Pharma to acquire PellePharm.
Targeted Oncology
- Infigratinib – FGFR1-3 inhibitor for FGFR+ cancer: Received FDA Fast Track Designation in adults with first-line advanced or metastatic cholangiocarcinoma and Orphan Drug Designation for the treatment of cholangiocarcinoma, and initiated Phase 3 study in advanced 1L cholangiocarcinoma as a first-line therapy (PROOF trial, NCT03773302).
- BBP-398 – SHP2 inhibitor for treatment-resistant cancer: Presented data (link to poster) highlighting the discovery and preclinical activity of our potent and selective SHP2 inhibitor potentially both as monotherapy and in combination with approved agents.
Gene Therapy
- BBP-812 – Gene therapy candidate for Canavan disease: Opened a natural history study in Canavan disease (treatcanavan.com) and presented preclinical data (link to poster) demonstrating intravenous (IV) dosing of BridgeBio’s experimental therapy for Canavan disease (BBP-812) achieved broad central nervous system delivery.
- BBP-631 – Gene therapy candidate for CAH: Presented preclinical update (link to poster) for gene therapy candidate BBP-631 in congenital adrenal hyperplasia (CAH) due to 21-hydroxylase deficiency wherein IV dosing of non-human primates with BBP-631 resulted in durable delivery and expression of the gene product to the adrenal tissue.
Upcoming milestones:
- Anticipate disclosing additional new product candidates and filing multiple new INDs in 2020.
- BBP-589 – COL7A protein replacement therapy for recessive dystrophic epidermolysis bullosa: Plan to share topline data from the ongoing Phase 1/2 study (NCT03752905) in 2020.
- Low-dose infigratinib – FGFR1-3 inhibitor for achondroplasia: Anticipate dosing of first child in Phase 2 dose-ranging study (PROPEL2) in the coming months.
- BBP-265 (AG10) – TTR stabilizer for ATTR: On track to complete enrollment in the Phase 3 study of AG10 in ATTR-CM in 2H20 (ATTRibute-CM). A Phase 3 study of AG10 in ATTR-PN (ATTRibute-PN) is on track to begin in 1H20.
- Encaleret – Calcium sensing receptor antagonist for autosomal dominant hypocalcemia type 1 (ADH1): Phase 2b ready.
- Infigratinib – FGFR1-3 inhibitor for FGFR+ cancer: Plan to present updated results at a major oncology meeting in 2020. Remain on track to submit new drug application for FDA approval in cholangiocarcinoma in 2020. Plan to dose first patients in a Phase 3 study in FGFR3+ adjuvant urothelial carcinoma (NCT04197986) and a Phase 2 tumor-agnostic study in patients with fusions or activating mutations in the FGFR1, 2 or 3 genes in 2020.
Organizational Growth:
- Ronald J. Daniels, board member: President of Johns Hopkins University joined board of directors.
- Jennifer Cook, board member and senior advisor: Genentech veteran who led Roche Pharma’s European commercial business joined board of directors and serves as a special advisor to BridgeBio.
- Eli Wallace, chief scientific officer in residence for oncology: Medicinal chemist and strategic executive leader who oversaw entire research organizations at companies such as Peloton Therapeutics and Array BioPharma joined BridgeBio to lead oncology research.
Fourth quarter and full-year 2019 financial results:
Cash, Cash Equivalents and Marketable Securities
Cash, cash equivalents and marketable securities, excluding restricted cash, totaled $577.1 million as of December 31, 2019 compared with $436.1 million at December 31, 2018.
License revenue
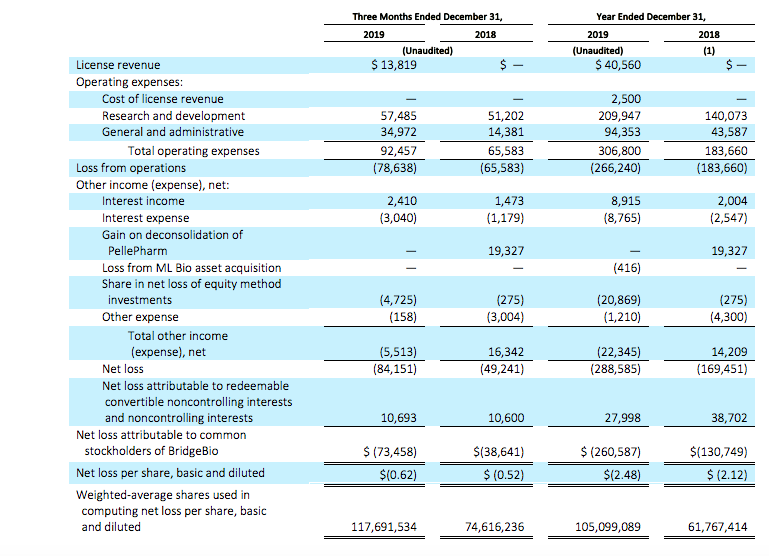
License revenue for the fourth quarter of 2019 was $13.8 million and for the full year 2019 was $40.6 million. The full year 2019 revenue was comprised mainly of the upfront payment recognized by our subsidiary company, Eidos Therapeutics, Inc. upon execution of its License Agreement with Alexion Pharmaceuticals, Inc.
Operating Expenses
Operating expenses for the fourth quarter of 2019 were $92.5 million, as compared to $65.6 million for the same period in the prior year. The increase in operating expenses of $26.9 million was primarily attributable to increase in headcount and external-related costs to support the growth of our operations.
Full year 2019 operating expenses were $306.8 million, as compared to $183.7 million for the full year in 2018. The increase of $123.1 million was mainly due to the increase in external-related costs to support the progression in our research and development programs and growth of our operations as well as increase in headcount.
Other Income (Expense), Net
Other income (expense), net for the fourth quarter of 2019 was ($5.5) million, as compared to $16.3 million for the same period in 2018. The change of $21.8 million was primarily attributable to a gain of $19.3 million that we recognized on deconsolidation of our controlled variable interest entity, PellePharm, Inc. in 2018.
Other income (expense), net for the full year 2019 was ($22.3) million, as compared to $14.2 million for the full year in 2018. The change of $36.5 million was primarily attributable to an increase in our net loss of equity method investments by $20.9 million in 2019 and the gain of $19.3 million on deconsolidation of our controlled variable interest entities, PellePharm, Inc.
BRIDGEBIO PHARMA, INC.
Condensed Consolidated Statements of Operations
(in thousands, except shares and per share amounts)
BRIDGEBIO PHARMA, INC.
Condensed Consolidated Balance Sheets
(In thousands)

(1) Upon the closing of our IPO on July 1, 2019, we completed a reorganization (the “Reorganization”), whereby all unitholders of BridgeBio Pharma LLC, our predecessor entity (“BBP LLC”) exchanged their units for shares of common stock of BridgeBio Pharma, Inc. (the “Corporation”), and BBP LLC became a wholly-owned subsidiary of the Corporation. Subsequent to the Reorganization, as the sole managing member, BridgeBio operates and controls all of BBP LLC’s businesses and affairs. The condensed consolidated financial statements as of and for the year ended December 31, 2018 are derived from the audited consolidated financial statements as of that date and were retroactively adjusted, including shares and per share amounts, as a result of the Reorganization. See the BridgeBio Pharma, Inc. Registration Statement on Form S-1 (File No. 333-231759) for additional details.
(2) December 31, 2019 amount includes long-term marketable securities of $31.1 million.
About BridgeBio Pharma
BridgeBio is a team of experienced drug discoverers, developers and innovators working to create life-altering medicines that target well-characterized genetic diseases at their source. BridgeBio was founded in 2015 to identify and advance transformative medicines to treat patients who suffer from Mendelian diseases, which are diseases that arise from defects in a single gene, and cancers with clear genetic drivers. BridgeBio’s pipeline of over 20 development programs includes product candidates ranging from early discovery to late-stage development. For more information, visit bridgebio.com.
BridgeBio Pharma Forward-Looking Statements
This press release contains forward-looking statements. Statements we make in this press release may include statements that are not historical facts and are considered forward-looking within the meaning of Section 27A of the Securities Act of 1933, as amended (the “Securities Act”), and Section 21E of the Securities Exchange Act of 1934, as amended (the “Exchange Act”), which are usually identified by the use of words such as “anticipates,” “believes,” “estimates,” “expects,” “intends,” “may,” “plans,” “projects,” “seeks,” “should,” “will,” and variations of such words or similar expressions. We intend these forward-looking statements to be covered by the safe harbor provisions for forward-looking statements contained in Section 27A of the Securities Act and Section 21E of the Exchange Act and are making this statement for purposes of complying with those safe harbor provisions. These forward-looking statements, including statements relating to expectations, plans and prospects regarding the preclinical and clinical development plans, clinical trial designs, clinical and therapeutic potential, and strategy of BridgeBio’s product candidates, including, but not limited to, the number of potential medicines in our portfolio, our plans to file an NDA for infigratinib in cholangiocarcinoma and multiple INDs for new product candidates in 2020, the initiation of our planned Phase 2/3 trial of Zuretinol, the availability of topline data from our Phase 1/2 study of BBP-589, the anticipated dosing of the first child in our Phase 2 dose-ranging study of infigratinib in achondroplasia, the completion of enrollment in our ongoing Phase 3 study of BBP-265 in ATTR-CM and the commencement of our planned Phase 3 study of BBP-265 in ATTR-PN, the availability of updated results for infigratinib and our plans for further clinical development of infigratinib in FGFR+ cancer, and the timing of these events, reflect our current views about our plans, intentions, expectations, strategies and prospects, which are based on the information currently available to us and on assumptions we have made. Although we believe that our plans, intentions, expectations, strategies and prospects as reflected in or suggested by those forward-looking statements are reasonable, we can give no assurance that the plans, intentions, expectations or strategies will be attained or achieved. Furthermore, actual results may differ materially from those described in the forward-looking statements and will be affected by a number of risks, uncertainties and assumptions, including, but not limited to, those risks set forth in the Risk Factors section of our most recent quarterly or annual periodic report filed with the SEC and our other SEC filings. Moreover, BridgeBio operates in a very competitive and rapidly changing environment in which new risks emerge from time to time. These forward-looking statements are based upon the current expectations and beliefs of BridgeBio’s management as of the date of this release, and are subject to certain risks and uncertainties that could cause actual results to differ materially from those described in the forward-looking statements. Except as required by applicable law, we assume no obligation to update publicly any forward-looking statements, whether as a result of new information, future events or otherwise.
Media Contact:
Grace Rauh, BridgeBio Pharma
[email protected]
917-232-5478
Investor Contact:
John Grimaldi, Burns McClellan
[email protected]
212-213-0006 x362
Source: BridgeBio Pharma